Inflammation that is aging your body from the inside and causing cognitive decline, joint pain, loss of muscle mass, and more is called inflammaging. Acute inflammation is necessary for healing. When it does its job, it retreats. Inflammaging is the opposite. It is chronic, systemic, low-grade, and self-sustaining. There is no wound to heal, no pathogen to defeat. Instead, your immune system stays partially activated indefinitely
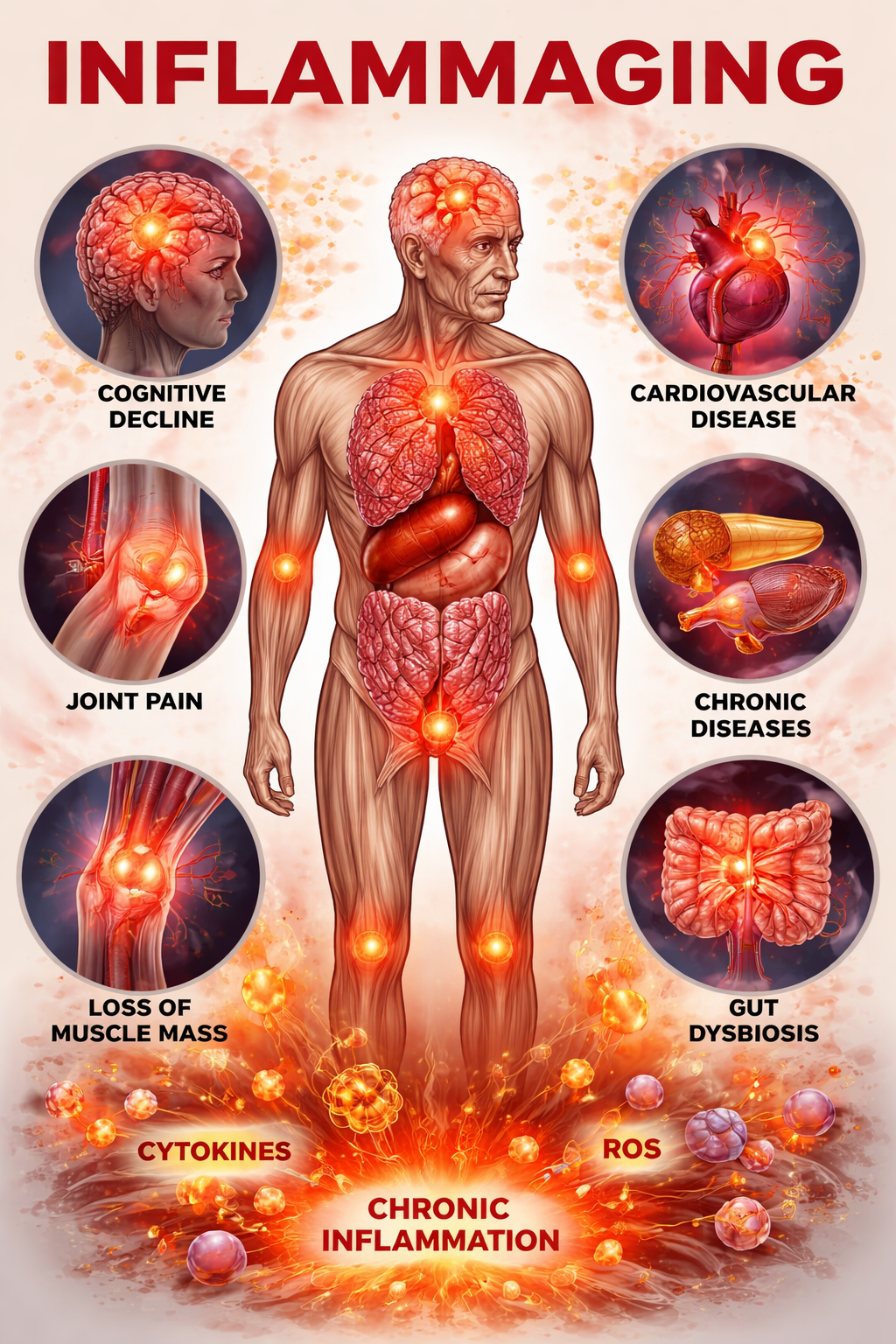
Imagine a fire burning inside you — not the kind you feel, but the kind that slowly consumes. It has no fever, no redness, no swelling you can see. It runs below the threshold of symptoms for years, sometimes decades, quietly eroding the tissues, vessels, and cells that keep you young. Scientists call it inflammaging — a portmanteau of inflammation and aging — and it is widely recognized as a key biological process associated with many major age-related diseases: heart disease, Alzheimer’s, type 2 diabetes, cancer, sarcopenia, and more.
The term was first coined in 2000 by Italian immunologist Claudio Franceschi, and the science around it has expanded significantly since. Inflammaging is increasingly considered a core feature (or “hallmark-associated process”) of aging, though it is more accurately described as a contributor and amplifier rather than a single root cause of all age-related disease. The good news: a large body of research shows that regular physical activity — including brief, higher-intensity bouts — can help modulate chronic inflammation.
Root Cause
Inflammation is involved in multiple leading causes of death globally, though it is not a single independent “root cause” category
Participants
Covered across large meta-analyses examining exercise and systemic inflammation (aggregated samples vary by analysis)
of Older Adults
Estimated to show elevated inflammatory markers with age (varies widely depending on biomarker and cutoff used)
PART ONE: UNDERSTANDING INFLAMMAGING
What Is Inflammaging — and Why Should You Care?
Acute inflammation is your friend. When you cut your finger or fight a virus, your immune system surges into action, deploying cytokines and immune cells to repair damage and eliminate threats. It is fast, targeted, and — critically — it resolves. The inflammation burns hot, does its job, and retreats.
Inflammaging is the opposite of all that. It is chronic, systemic, low-grade, and self-sustaining. There is no wound to heal, no pathogen to defeat. Instead, your immune system stays partially activated indefinitely, releasing a low steady stream of pro-inflammatory signaling molecules — primarily interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-?), C-reactive protein (CRP), and interleukin-1? (IL-1?) — without ever resolving the response.
What feeds this slow burn? Researchers have identified several converging mechanisms:
As cells age, they enter a state of irreversible growth arrest. Rather than dying cleanly, these “zombie cells” linger and release a cocktail of pro-inflammatory signals known as the Senescence-Associated Secretory Phenotype (SASP) — continuously fueling systemic inflammation.
Mitochondrial Dysfunction
Aging mitochondria become leaky, releasing fragments of mitochondrial DNA into the cytoplasm. The body mistakes these fragments for bacterial DNA and triggers an immune alarm via the cGAS-STING pathway.
Visceral Adipose Tissue
Abdominal fat is metabolically active tissue packed with inflammatory macrophages. As visceral fat expands with age, it becomes a continuous source of IL-6, TNF-?, and other pro-inflammatory cytokines — feeding systemic inflammaging directly.
NLRP3 Inflammasome
This intracellular immune sensor becomes increasingly hair-triggered with age, activating in response to metabolic stress and releasing IL-1? even in the absence of infection.
Immunosenescence
The immune system itself ages, losing the ability to clear senescent cells, resolve inflammatory signals, or mount effective anti-inflammatory responses — creating a vicious cycle where inflammation accelerates immune aging.
Gut Microbiome Dysbiosis
Age-related changes in gut bacteria and increased intestinal permeability allow bacterial fragments (lipopolysaccharides) to leak into circulation, triggering chronic low-grade immune activation.
— Nature Reviews Cardiology (Libby et al.)
The Disease Downstream of the Flame
Why does inflammaging matter clinically? Because chronic inflammation is strongly associated with multiple diseases:
Cardiovascular Disease. Elevated CRP and IL-6 are associated with endothelial dysfunction and atherosclerosis.
Neurodegenerative Disease. Chronic neuroinflammation contributes to Alzheimer’s and Parkinson’s disease, though causality is complex and bidirectional.
Metabolic Disease. TNF-? and IL-6 contribute to insulin resistance, though they are part of a broader metabolic network.
Sarcopenia and Frailty. Elevated inflammatory markers are associated with muscle loss and functional decline.
Cancer. Chronic inflammation is a known enabling factor in tumor development, though mechanisms vary widely by cancer type.
THE BIOMARKER BASICS
Inflammaging is measurable. Ask your doctor about these key inflammatory markers on your next blood panel:
- hsCRP (high-sensitivity C-reactive protein): Target <1.0 mg/L (low risk); >3.0 mg/L = high inflammatory risk
- IL-6: Chronically elevated levels predict cardiovascular risk and functional decline
- TNF-?: A key driver of insulin resistance and muscle catabolism
- Fibrinogen: An acute-phase protein that rises with systemic inflammation; tracks cardiovascular risk
PART TWO: YOUR MUSCLES AS MEDICINE
The Myokine Revolution
For decades, exercise was viewed primarily as a calorie-burning, heart-strengthening activity. We now know it is far more than that. Skeletal muscle — the body’s largest organ by mass — is also a potent endocrine organ. When you contract your muscles, they release a cascade of signaling proteins called myokines. And many of these myokines are directly, powerfully anti-inflammatory.
The discovery that changed everything came in 2000, when Danish physiologist Bente Klarlund Pedersen identified interleukin-6 (IL-6) as the first myokine. What was surprising: IL-6 released from contracting muscle tissue behaves completely differently from IL-6 released during infection or disease. Muscle-derived IL-6 is actually anti-inflammatory. It stimulates production of IL-10 and IL-1 receptor antagonist (IL-1ra) — both of which suppress the pro-inflammatory cascade — while simultaneously inhibiting TNF-? production.
This distinction matters enormously. The same cytokine that, when elevated chronically due to disease, is a marker of inflammaging — when produced acutely by muscle contraction, functions as an anti-inflammatory agent. Exercise does not merely reduce inflammation passively. It actively deploys your muscles as a biological pharmacy.
The Evidence: What Exercise Does to Your Inflammatory Markers
The science here is now robust. A landmark 2025 meta-meta-analysis — a systematic review of 25 meta-analyses and systematic reviews, encompassing a total of 30,017 participants — found that exercise interventions produced significant reductions across all three primary markers of chronic systemic inflammation:
? Pooled effect
?0.38
CRP is the primary clinical marker for cardiovascular inflammatory risk — levels above 3 mg/L substantially elevate heart attack risk independent of cholesterol
IL-6
? Pooled effect
?0.47
Chronically elevated IL-6 drives endothelial dysfunction, insulin resistance, muscle wasting, and neuroinflammation
TNF-?
? Pooled effect
?0.43
TNF-? is the primary cytokine responsible for insulin resistance, and a key driver of the senescence-associated secretory phenotype (SASP)
A companion meta-analysis specifically in older adults (Oxford University, 2025) confirmed that circulating concentrations of CRP, TNF-?, and IL-6 all decrease in response to physical exercise interventions in elderly populations — precisely the group most burdened by inflammaging.
A separate network meta-analysis of 123 randomized controlled trials examined which exercise types were most effective for different inflammatory markers in people with overweight or obesity. The findings were specific and actionable: HIIT (high-intensity interval training) produced the largest effect sizes for reducing IL-6, TNF-?, and increasing anti-inflammatory IL-10, while aerobic exercise was most effective for CRP and adiponectin. Combined training had the broadest benefits.
The Dose Question: How Little Exercise Is Enough?
Here is the question that matters most for busy people: does the exercise have to be prolonged to produce anti-inflammatory effects? The research increasingly says no.
A 2023 Frontiers in Psychology meta-analysis (38 RCTs, 2,557 healthy participants) found that long-term exercise training significantly reduced IL-6, CRP, and TNF-?, with subgroup analysis revealing that moderate-intensity training showed robust anti-inflammatory effects — and, crucially, that the reduction in CRP was actually weakened at very high exercise intensities. This suggests that brief-but-vigorous microdose sessions sit in an anti-inflammatory sweet spot.
A separate meta-analysis in elderly adults showed that resistance training lasting as few as 8 weeks — at vigorous intensity, with as few as 8 exercises, performed just 2 times per week — produced the largest CRP reductions of any protocol tested. Both moderate and vigorous exercise can reduce inflammation; optimal results depend on consistency and total volume.
The Five Biological Pathways Exercise Activates Against Inflammaging
Muscle contraction triggers release of IL-6 (muscle-derived, anti-inflammatory), IL-10, IL-15, irisin, and BDNF. These myokines suppress TNF-? production, upregulate anti-inflammatory pathways, and modulate immune cell activity across the entire body.
Senescent Cell Clearance
Exercise is associated with reductions in markers linked to cellular senescence and improved cellular function. While animal and early human studies are promising, direct evidence of large-scale senescent cell clearance in humans is still emerging.
Visceral Fat Mobilization
Even brief vigorous exercise preferentially mobilizes visceral adipose tissue — the single largest source of chronic inflammatory cytokines. As visceral fat shrinks, the macrophage population within it shifts from pro-inflammatory M1 phenotype to anti-inflammatory M2, dramatically reducing the tissue’s contribution to systemic inflammation.
Telomere Protection & Anti-Senescence
Exercise upregulates telomere-stabilizing proteins and increases telomerase activity in circulating leukocytes and vascular wall cells. The underlying mechanism involves increased endothelial nitric oxide (NO), which synergizes with telomerase to protect against cellular senescence — even after as little as 3 weeks of voluntary exercise in animal models.
TLR Suppression
Physical inactivity is associated with heightened activation of Toll-like receptors (TLRs), the immune sensors that detect damage signals and initiate inflammatory cascades. Regular moderate-to-vigorous exercise reduces TLR expression in immune cells, lowering their sensitivity to inflammatory triggers — essentially cooling down the immune alarm system.
PART THREE: THE ZOMBIE CELL PROBLEM — AND HOW EXERCISE SOLVES IT
Senescent Cells: The Hidden Architecture of Inflammaging
Of all the drivers of inflammaging, senescent cells may be the most consequential — and the most exciting from a therapeutic standpoint. When cells suffer DNA damage, telomere shortening, or oncogenic stress, many enter a state of permanent cell cycle arrest. They stop dividing, which initially serves a protective anti-tumor function. But rather than dying through apoptosis, they linger — and they are anything but quiet.
Senescent cells continuously secrete the SASP: a complex mix of pro-inflammatory cytokines (IL-1?, IL-6, IL-8, TNF-?), chemokines, matrix-remodeling enzymes, and growth factors. Locally, SASP signals disrupt tissue structure and function. Systemically, they elevate inflammatory markers throughout the body. And — in a particularly cruel feedback loop — SASP signals can induce neighboring healthy cells to become senescent themselves, spreading the dysfunction.
Senescent cells are rare in young tissues because the immune system efficiently clears them. With age, both the rate of senescent cell formation increases and the immune system’s capacity to clear them declines. The result is accumulation — and the accumulation correlates directly with disease burden, frailty, and mortality.
Exercise as a Senolytic — The Evidence
Can exercise actually clear senescent cells? The data is compelling:
Exercise Reduces Biomarkers of Cellular Senescence in Humans
A landmark study published in Aging Cell followed older adults through a 12-week progressive strength and endurance training intervention (2 days/week). Results:
- Expression of senescence markers p16 and p21 in circulating T cells was significantly and consistently reduced
- Components of the cGAS-STING pathway (including IFN-? and TNF-?) — which triggers inflammation and reinforces senescence — were reduced
- Circulating SASP proteins including myeloperoxidase and serpin E1 (PAI-1) were lowered
- The baseline SASP index predicted who responded to the exercise intervention — individuals with higher senescent cell burden showed the greatest improvements in physical function
Source: Aging Cell (Dungan et al.); DOI: 10.1111/acel.13415
A separate systematic review on exercise as a senolytic medicine confirmed that exercise can reduce markers of senescent cells in healthy humans, including the canonical CDKI p16 and p21 — the same targets pursued by pharmaceutical senolytic drugs currently in clinical trials. Notably, HIIT specifically and significantly decreases markers of senescent cells, with the largest effects seen in individuals with higher baseline senescence burden — exactly the population that most needs it.
The mechanism involves the acute inflammatory response triggered by vigorous exercise. When you perform high-intensity activity, macrophages and NK cells are mobilized and activated. This immune surge — transient, purposeful, and self-resolving — appears to enhance the immune system’s capacity to identify and clear senescent tissue. In contrast to the smoldering fire of inflammaging, exercise creates a controlled, targeted, beneficial inflammatory pulse that burns out the debris.
PART FOUR: THE MICRODOSE PRESCRIPTION
Brief, Vigorous, and Repeated — The Anti-Inflammaging Formula
What does the research tell us about the optimal anti-inflammaging exercise strategy? Three principles emerge consistently from the literature:
Intensity matters more than duration. HIIT and vigorous-intensity resistance training show the largest effect sizes for reducing IL-6, TNF-?, and clearing senescent cell markers. This is why microdose fitness — short, high-effort bursts spread through the day — is physiologically well-suited for anti-inflammaging.
Frequency and consistency matter more than single long sessions. The anti-inflammatory adaptation is a chronic response built through regular repeated stimulation of myokine release. Doing 10 minutes of vigorous activity five days a week creates more sustained anti-inflammatory signaling than one 50-minute weekend session.
Muscle mass is protective. Resistance training that builds and preserves muscle tissue is particularly important for older adults. Greater muscle mass means greater myokine production capacity — a larger biological anti-inflammatory pharmacy running continuously.
The Microdose Anti-Inflammaging Menu
Each of the following can be performed in 2–10 minutes at home, at work, or anywhere throughout your day. The goal is to generate meaningful muscle contraction and brief cardiovascular elevation — the two triggers for maximal myokine release:
Stair Sprints
2–4 flights, maximum effort, 2–3x/day
Triggers high-volume myokine release including IL-6 (anti-inflammatory), IL-15, and irisin; activates lower-body fast-twitch fibers most associated with IL-10 induction
Jump Squats
3 sets of 10, 30-sec rest
Intense eccentric + concentric load activates the acute inflammatory pulse needed to trigger subsequent SASP clearance; builds quadricep and glute mass for sustained myokine capacity
Kettlebell Swings
3 sets of 15, any weight
Full-body posterior chain activation; combines cardiovascular stimulus with resistance for combined aerobic + resistance anti-inflammatory effect shown to be most effective in meta-analyses
Resistance Band Work
2 sets of 15, upper + lower
Accessible form of resistance training shown to reduce CRP in elderly populations; particularly important for older adults who cannot perform high-impact moves
Burpees
10 reps × 3 sets
Combines full-body resistance with cardiorespiratory stress; produces large acute myokine surge; especially effective for visceral fat mobilization
Brisk Walk Intervals
5-min walk, 1-min hard push × 3
For those new to exercise or managing chronic conditions; 2025 ACC guidelines endorse ?75 min/week vigorous aerobic for reducing chronic low-grade inflammation.
Building Your Anti-Inflammaging Protocol
RECOMMENDED MICRODOSE SCHEDULE
Build anti-inflammatory protection through consistency, not duration:
- Monday / Wednesday / Friday — Resistance focus: Jump squats, kettlebell swings, or resistance band circuit (8–12 minutes). These sessions build myokine-producing muscle mass and clear senescent cells.
- Tuesday / Thursday — Cardio spikes: 2–3 sets of stair sprints, brisk walk intervals, or 10-minute effort bursts. These produce the sharpest myokine cascade and the most visceral fat mobilization.
- Saturday — Combined: 20–30 minutes mixing aerobic and resistance. Meta-analyses confirm combined training produces the broadest anti-inflammatory effects.
- Sunday — Active recovery: gentle walking, stretching. Even light movement supports anti-inflammatory microbiome health and insulin sensitivity.
What Not to Do: The Anti-Inflammatory Saboteurs
Research has identified several common behaviors that blunt the anti-inflammatory response to exercise. Awareness of these can help you protect the gains you make:
NSAIDs immediately post-exercise. Taking ibuprofen or other NSAIDs right after exercise suppresses the acute inflammatory response needed to clear senescent cells and trigger anti-inflammatory adaptation. Reserve NSAIDs for genuine injury, not routine post-workout use.
Cold water immersion immediately post-exercise. Ice baths and cold plunges suppress the acute inflammatory pulse that drives the beneficial cellular adaptations from HIIT. If you use cold exposure, separate it from intense exercise by several hours.
Excessive antioxidant supplementation. High-dose antioxidants (Vitamins C and E) taken around workouts can blunt the exercise-induced signaling cascade that drives long-term anti-inflammatory adaptation. Food-based antioxidants are fine; megadose supplements peri-workout may not be.
Chronic sedentary behavior between sessions. Extended sitting between microdose sessions is independently associated with elevated inflammatory markers. The goal is to scatter movement throughout the day, not to offset all-day sitting with one exercise burst.
PART FIVE: THE BIGGER PICTURE
Exercise as Preventive Anti-Inflammatory Medicine
What emerges from the full body of research is a striking reframing of exercise: not merely as a strategy for fitness, weight management, or cardiovascular health, but as the most evidence-based, broadly accessible anti-inflammatory therapy available to humans.
The 2025 American College of Cardiology Scientific Statement explicitly recommends at least 75 minutes per week of vigorous aerobic exercise as a lifestyle strategy to reduce chronic low-grade inflammation and lower atherosclerotic cardiovascular disease risk. The evidence base for this recommendation includes decades of randomized controlled trials and multiple layers of meta-analysis.
The microdose framework makes this achievable for people who cannot commit to prolonged gym sessions. Five 15-minute sessions of vigorous activity — three resistance and two cardio — exceeds the recommended threshold while fitting into any schedule, requiring no equipment beyond bodyweight and a staircase.
The Timeline: When Does Anti-Inflammatory Benefit Begin?
WHAT TO EXPECT — A REALISTIC TIMELINE
Build anti-inflammatory protection through consistency, not duration:
- Session 1–3: Acute myokine release occurs with every session. IL-6, IL-10, and IL-1ra surge in the hours following intense exercise, providing an immediate anti-inflammatory effect that peaks 1–3 hours post-workout.
- Weeks 1–4: First measurable reductions in circulating CRP and TNF-? typically appear within 4 weeks of consistent training. Senescent cell marker expression begins to decline in peripheral blood T cells.
- Weeks 8–12: Significant reductions in CRP, IL-6, and TNF-? reach statistical significance in most RCTs. Studies confirm resistance training conducted for more than 12 weeks and at vigorous intensity produces the largest CRP effect sizes, including in adults over 70.
- Month 6+: Muscle mass gains compound the myokine effect. As muscle tissue grows, the body’s capacity for anti-inflammatory myokine production increases with it. Visceral fat reduction further removes a primary source of systemic inflammation.
*Timelines are approximate, not guaranteed
THE BOTTOM LINE
Inflammaging is real, measurable, and consequential. It is the biological thread connecting heart disease, dementia, diabetes, cancer, and frailty. It accumulates quietly for decades, driven by zombie cells, toxic fat tissue, aging mitochondria, and an immune system that has lost its off switch.
And your muscles are wired to fight it. Every time you contract skeletal muscle with intensity, you trigger a cascade of anti-inflammatory myokines, mobilize visceral fat, activate immune senescent cell clearance, and stabilize the very telomeres that protect your cells from aging. The effects are dose-dependent, cumulative, and scientifically confirmed in tens of thousands of research participants.
Microdose fitness is not a compromise. It is not “good enough” exercise. For the specific goal of combating inflammaging, brief vigorous sessions repeated consistently throughout the week may be the optimal strategy — producing the acute inflammatory pulse and myokine surge that drives the deepest anti-inflammatory adaptation, without the recovery cost of prolonged training.
The fire within is real. But so is your ability to fight it. Start with two flights of stairs today.
MEDICAL DISCLAIMER
This article is for informational and educational purposes only. It is not intended to constitute medical advice, diagnosis, or treatment. Consult a qualified healthcare provider before beginning any new exercise program, particularly if you have existing health conditions, are taking medications, or are experiencing symptoms of disease. The studies cited represent current research findings but individual results will vary.
Scientific References
- Liberale L, et al. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nature Reviews Cardiology (2020). PMC6146930
- Colon & colleagues. Inflammation and aging-related disease: A transdisciplinary inflammaging framework. GeroScience (2025). PMC11872841
- Jiang et al. Global research trends in inflammaging from 2005 to 2024: a bibliometric analysis. Frontiers in Aging (2025). DOI: 10.3389/fragi.2025.1554186
- Pedersen BK. The anti-inflammatory effect of exercise. PubMed Review (2005). PMID: 15772055
- Ringleb M et al. Beyond muscles: Investigating immunoregulatory myokines in acute resistance exercise. The FASEB Journal (2024). DOI: 10.1096/fj.202301619R
- Karavelioglu B et al. The impact of exercise on chronic systemic inflammation. Sport Sciences for Health (2025). DOI: 10.1007/s11332-025-01445-3
- Chen C et al. Effects of various exercise types on inflammatory response in overweight/obese individuals. Network Meta-Analysis (2024).
- Mathot E et al. Physical interventions in older adults on inflammatory markers. Innovation in Aging (Oxford, 2025). DOI: 10.1093/geroni/igaf072
- Yang YR et al. Effects of Resistance Training on C-Reactive Protein and Inflammatory Cytokines in Elderly Adults. International Journal of Environmental Research and Public Health (2022). MDPI
- Dungan et al. Exercise reduces circulating biomarkers of cellular senescence in humans. Aging Cell (2022). PMC8282238
- Kehler DS et al. Exercise Counters the Age-Related Accumulation of Senescent Cells. PMC Review (2022). PMC9680689
- Hashida R et al. Is exercise a senolytic medicine? A systematic review. PubMed (2021). PMID: 33378138
- Werner C et al. Physical Exercise Prevents Cellular Senescence in Circulating Leukocytes and in the Vessel Wall. Circulation (2009). AHA Journals
- Ridker PM et al. Inflammation and Cardiovascular Disease: 2025 ACC Scientific Statement. JACC (2025). DOI: 10.1016/j.jacc.2025.08.047
- Zhong Z et al. Inflammation and aging: signaling pathways and intervention therapies. Signal Transduction and Targeted Therapy (2023). Nature